Chemistry and classes of Bisphosphonates
All bisphosphonate drugs share a common P-C-P "backbone":
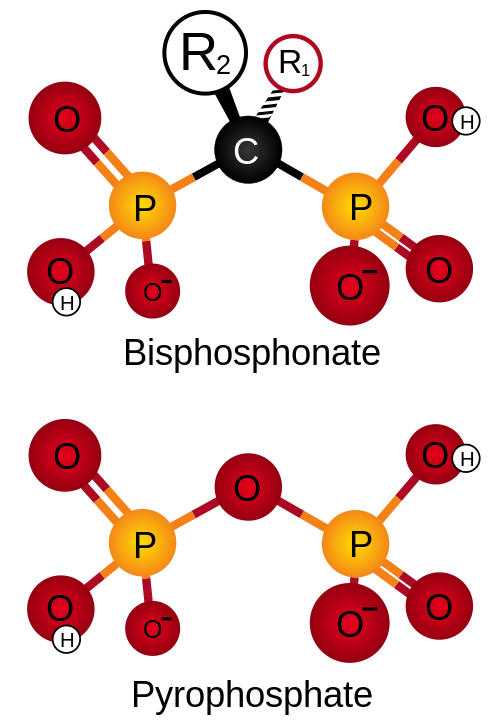
Basic structure of a bisphosphonate on top. To compare the structure of pyrophosphate below. Note the similarity in structure. The two PO3 (phosphonate) groups covalently linked to carbon determine both the name "bisphosphonate" and the function of the drugs.
The long side-chain (R2 in the diagram) determines the chemical properties, the mode of action and the strength of bisphosphonate drugs. The short side-chain (R1), often called the 'hook', mainly influences chemical properties and pharmacokinetics.
Structural similarity between a bisphosphonate group and a pyrophosphate molecule
Classification of Bisphosphonates:
a. Non-nitrogenous:
Non-N-containing bisphosphonates:
- Etidronate (Didronel) - 1 (potency relative to that of etidronate)
- Clodronate (Bonefos, Loron) - 10
- Tiludronate (Skelid) - 10
b. Nitrogenous N-containing bisphosphonates:
- Pamidronate (APD, Aredia) - 100
- Neridronate - 100
- Olpadronate - 500
- Alendronate (Fosamax) - 500
- Ibandronate (Boniva) - 1000
- Risedronate (Actonel) - 2000
- Zoledronate (Zometa, Aclasta) - 10000
Pharmacokinetics
Of the bisphosphonate that is resorbed (from oral preparation) or infused (for intravenous drugs), about 50% is excreted unchanged by the kidney. The remainder has a very high affinity for bone tissue, and is rapidly adsorbed onto the bone surface.
Mechanism of action:
- Non- nitrogenous Bisphosphonates: The non-nitrogenous bisphosphonates are metabolized in the cell to compounds that replace the terminal pyrophosphate moiety of ATP, forming a nonfunctional molecule that competes with adenosine triphosphate (ATP) in the cellular energy metabolism. The osteoclast initiates apoptosis and dies, leading to an overall decrease in the breakdown of bone.
- Nitrogenous bisphosphonates: Nitrogenous bisphosphonates act on bone metabolism by binding and blocking the enzyme farnesyl diphosphate synthase (FPPS) in the HMG-CoA reductase pathway (also known as the mevalonate pathway).

Disruption of the HMG CoA-reductase pathway at the level of FPPS prevents the formation of two metabolites farnesol and geranylgeraniol that are essential for connecting some small proteins to the cell membrane. This phenomenon is known as prenylation, and is important for proper sub-cellular protein trafficking (see "lipid-anchored protein" for the principles of this phenomenon).[6]
While inhibition of protein prenylation may affect many proteins found in an osteoclast, disruption to the lipid modification of Ras, Rho, Rac proteins has been speculated to underlie the effects of bisphosphonates. These proteins can affect osteoclastogenesis, cell survival, and cytoskeleton dynamics. In particular, the cytoskeleton is vital for maintaining the "ruffled border" that is required for contact between a resorbing osteoclast and a bone surface.
Statins are another class of drugs that inhibit the HMG-CoA reductase pathway.
Unlike bisphosphonates, statins do not bind to bone surfaces with high affinity, and are, thus, not specific for bone. Nevertheless, some studies have reported a decreased rate of fracture (an indicator of osteoporosis) and/or an increased bone mineral density in statin users. The overall efficacy of statins in the treatment osteoporosis remains controversial.
Important Reference:
Drake MT, Clarke BL, Khosla S. Bisphosphonates: mechanism of action and role in clinical practice. Mayo Clin Proc. 2008 Sep;83(9):1032-45. Click Here for free full text of the article
Wikipedia: http://en.wikipedia.org/wiki/Bisphosphonate













 India-Orth
India-Orth